The network for creativity
Join 1.25M professional creatives like you
Connect with clients, get discovered, and run your business 100% commission-free
Creatives on Contra have earned over $150M and we are just getting started
Back to feedPost
#Oralsoliddosage
hashtag#pharmaceuticalosdmanufacturing
hashtag#Chantillyusajob
hashtag#opentnvisa
hashtag#hvacsystems
Hi All repost below !
Senior Manager, Engineering (Oral Solid Dosage)
Chantilly, Virginia, USA
Domain
Pharmaceuticals/Biotechnology/Clinical Research
Experience
10 - 25 Years
Location
USA - Chantilly
Open to give TN Visa
Key Responsibilities
Engineering Leadership
Lead and manage engineering teams supporting OSD manufacturing, packaging, utilities, and facility systems.
Provide technical direction for troubleshooting, equipment optimization, and process improvements.
Develop longterm engineering strategies that support operational excellence and asset reliability.
OSD Equipment & Process Support
Oversee installation, qualification, and maintenance of OSD equipment such as granulators, fluid bed dryers, tablet presses, capsule fillers, coaters, blenders, and packaging lines.
Ensure equipment operates at peak performance with minimal downtime.
Lead rootcause investigations and implement CAPA for equipment and process issues.
Experience in PM and Calibration of the equipment.
Facility Engineering & Utilities Management
Oversee operation and maintenance of HVAC systems, cleanrooms, compressed air, chilled water, boilers, vacuum systems, and other critical utilities.
Ensure environmental controls meet cGMP requirements for temperature, humidity, pressure differentials, and air quality.
Manage facility maintenance programs, preventive maintenance schedules, and reliability initiatives.
Support facility expansions, renovations, and infrastructure upgrades.
Ensure compliance with OSHA, environmental regulations, and internal safety standards.
Compliance & Quality
Ensure all engineering and facility operations comply with cGMP, FDA, EU, and internal quality standards.
Support audits, inspections, and documentation requirements.
Maintain engineering documentation, change controls, and validation protocols.
Safety & Risk Management
Champion a strong safety culture across engineering and operations.
Conduct risk assessments (FMEA, HAZOP) and implement engineering controls to ensure safe operations.
Identify opportunities for automation, modernization, and energy efficiency.
Team Development
Mentor and develop engineering and facility staff, fostering technical growth and leadership capability.
Build a collaborative, highperformance engineering organization.
Qualification Required:
Bachelor’s degree in mechanical, Chemical, Electrical Engineering, or related field.
12+ years of engineering experience in pharmaceutical OSD manufacturing.
Strong knowledge of facility engineering, including HVAC, clean utilities, building systems, and environmental controls.
Proven leadership experience managing engineering and facility teams.
Deep understanding of cGMP, validation, and regulatory requirements.
Subject line should be "Senior Manager, Engineering (Oral Solid Dosage)".Pls share text at Linkedin

linkedin.com
LinkedIn Login, Sign in | LinkedIn
Login to LinkedIn to keep in touch with people you know, share ideas, and build your career.
The network for creativity
Join 1.25M professional creatives like you
Connect with clients, get discovered, and run your business 100% commission-free
Creatives on Contra have earned over $150M and we are just getting started
Related posts
Title:
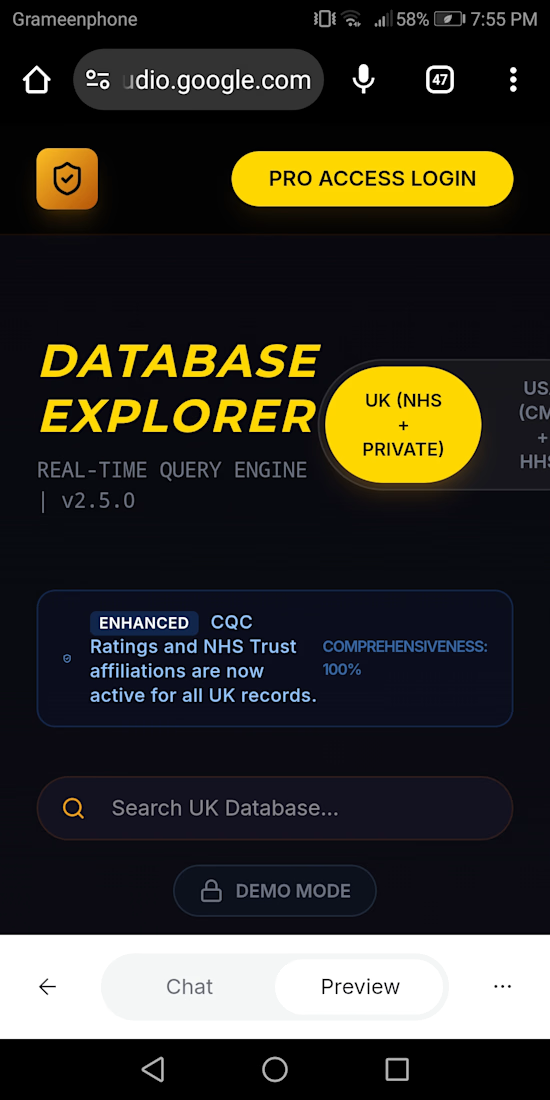
Healthcare Lead Intelligence Platform | UK & USA Hospitals
Short Description:
Searchable UK & USA hospital database for recruiters, suppliers, and healthcare research. Fast access to organized hospital records and lead discovery tools.
Link:
https://ai.studio/apps/828248e1-8f40-44be-8456-3820ac930c27�
Data AnalysisLead GenerationDatabase DevelopmentGoogle GeminiMicrosoft Copilot StudioGoogle AI Studio


Looking to hire
Senior Sales Manager – Industrial Cleaning Equipment Sales
Industry: Industrial Cleaning Equipment | Portable Washing Systems | Pressure Washer Solutions
Product Focus: Portable Washing Kit / Portable Cleaning Systems
Location: Bangalore (with extensive travel across India and export markets)
Role Type: Full-time | Individual Contributor / Strategic Sales Role
Role Summary
We are looking for an experienced Industrial Cleaning Equipment Sales Professional who has successfully sold pressure washers, cleaning equipment, portable washing systems, or industrial cleaning solutions to companies across multiple sectors in India and international markets.
This role is specifically designed for candidates who have direct experience in selling cleaning equipment to B2B customers such as automotive companies, industrial plants, facility management companies, equipment distributors, construction firms, and export buyers.
The selected candidate will lead the market expansion of our company's Portable Washing Kit, targeting multiple high-consumption sectors such as:
• Automotive service networks
• Industrial plants
• Fleet operators
• Construction companies
• Facility management firms
• Railways, Bus Depot/Stations and infrastructure companies
• Equipment distributors
• International buyers
The role requires someone who already understands the cleaning equipment ecosystem, knows how to identify bulk buyers, and can quickly generate high-value orders.
Key Responsibilities
Industrial Equipment Sales & Market Development
• Drive sales of portable washing kits and cleaning systems across multiple industrial sectors.
• Identify and approach companies currently using pressure washers, industrial cleaning equipment, and wash systems.
• Build relationships with purchase managers, plant heads, maintenance heads, and operations leaders.
• Develop high-volume sales opportunities with OEM service networks and industrial clients.
B2B Client Acquisition: Actively target companies that regularly use cleaning systems including:
• Automotive OEM workshops
• Vehicle service networks
• Logistics fleet operators
• Industrial manufacturing plants
• Construction companies
• Equipment rental companies
• Facility management companies
• Airports, railways,Bus depots/stations and infrastructure projects
Distributor & Channel Development
• Identify and onboard industrial equipment distributors and dealers.
• Build channel networks across key cities and industrial clusters.
• Establish long-term distributor partnerships to expand product reach.
Export Market Expansion
• Identify and connect with international buyers and distributors of cleaning equipment.
• Build export relationships in markets such as:
• Middle East
• Southeast Asia
• Africa
• Latin America
• Participate in global trade platforms and export outreach.
Sales Pipeline & Deal Management
• Generate and maintain a pipeline of high-value B2B prospects.
• Conduct product demonstrations and technical presentations.
• Prepare quotations, commercial proposals, and negotiations.
• Convert leads into confirmed orders.
Strategic Market Intelligence
• Track competitor pricing and product positioning in the cleaning equipment market.
• Identify high-demand sectors where portable washing systems can scale quickly.
• Provide market insights to help improve product positioning and sales strategy.
Required Experience
We are specifically looking for candidates who have already sold cleaning equipment or pressure washers.
Mandatory Experience
Candidates must have experience in at least one of the following industries:
• Industrial cleaning equipment
• Pressure washer systems
• Portable washing machines
• High-pressure cleaning systems
• Car wash equipment
• Industrial maintenance equipment
Sales Experience
• 4-6 years of B2B sales experience in cleaning equipment or industrial equipment.
• Proven experience selling products to industrial companies or service networks.
• Experience handling large orders, distributor networks, or export clients.
Required Skills
Industry Knowledge
• Strong understanding of pressure washer systems and cleaning equipment applications.
• Knowledge of industries that consume large volumes of cleaning equipment.
B2B Sales Skills
• Prospecting and client acquisition
• Distributor development
• Negotiation and deal closure
• Industrial client relationship management
Communication Skills
• Professional email and phone communication
• Ability to explain product benefits and ROI to industrial buyers
• Confidence presenting to decision-makers and procurement teams
Desired Qualifications
• Bachelor’s Degree (Engineering / Business / Marketing preferred)
• Sales or export certifications are an advantage
• Experience working with industrial distributors or equipment dealers
Key Performance Indicators (KPIs)
The success of this role will be measured through:
• Number of industrial clients acquired
• Distributor partnerships created
• Monthly sales revenue generated
• Number of export clients onboarded
• Conversion rate from leads to confirmed orders
• Expansion into new industry sectors
Why This Role Matters
Portable cleaning systems are becoming essential across multiple industries due to increasing demand for mobility, efficiency, and water-saving cleaning solutions.
This role will lead the market expansion of our portable washing systems across India and international markets.
The right candidate will directly contribute to:
• Rapid sales growth
• Expansion into new industry segments
• Building a nationwide distribution network
• Scaling export opportunities
What Makes This Role Exciting
● Sell a Product With Massive Industry Demand: Portable cleaning systems are required across automotive, construction, logistics, and industrial
Pls share resume at my Linkedin link : https://www.linkedin.com/in/technicalrecruitersuma
Trending
Claude
Claude has entered the design space. How are you using Claude Design?
Contra University
Learn from expert creatives how to earn more using next-gen AI tools.
creativeaiflow
Creative AI workflows are evolving. What tools do you use, and what are their strengths and weaknesses?
portfolioreview
The best portfolios tell a story, not just show a grid. Share yours for feedback.
freelancerlife
Freelancer life is wins, pivots, and everything in between. What’s yours right now?